Definite Shape Of A Solid
8.2: Solids and Liquids
- Folio ID
- 16101
- To describe the solid and liquid phases.
Solids and liquids are collectively called condensed phases because their particles are in virtual contact. The two states share little else, however.
Solids
In the solid state, the individual particles of a substance are in fixed positions with respect to each other because in that location is not enough thermal energy to overcome the intermolecular interactions between the particles. As a result, solids have a definite shape and volume. Most solids are hard, just some (like waxes) are relatively soft. Many solids composed of ions can also exist quite breakable.

Solids usually take their elective particles bundled in a regular, three-dimensional assortment of alternating positive and negative ions chosen a crystal. The effect of this regular arrangement of particles is sometimes visible macroscopically, as shown in Effigy \(\PageIndex{1}\). Some solids, especially those composed of large molecules, cannot hands organize their particles in such regular crystals and exist every bit amorphous (literally, "without course") solids. Glass is one example of an baggy solid.
Liquids
If the particles of a substance have enough free energy to partially overcome intermolecular interactions, so the particles tin move about each other while remaining in contact. This describes the liquid state. In a liquid, the particles are still in close contact, and then liquids have a definite volume. However, because the particles can move about each other rather freely, a liquid has no definite shape and takes a shape dictated by its container.

Gases
If the particles of a substance have plenty free energy to completely overcome intermolecular interactions, so the particles can separate from each other and movement about randomly in space. Like liquids, gases have no definite shape, only different solids and liquids, gases have no definite volume either.
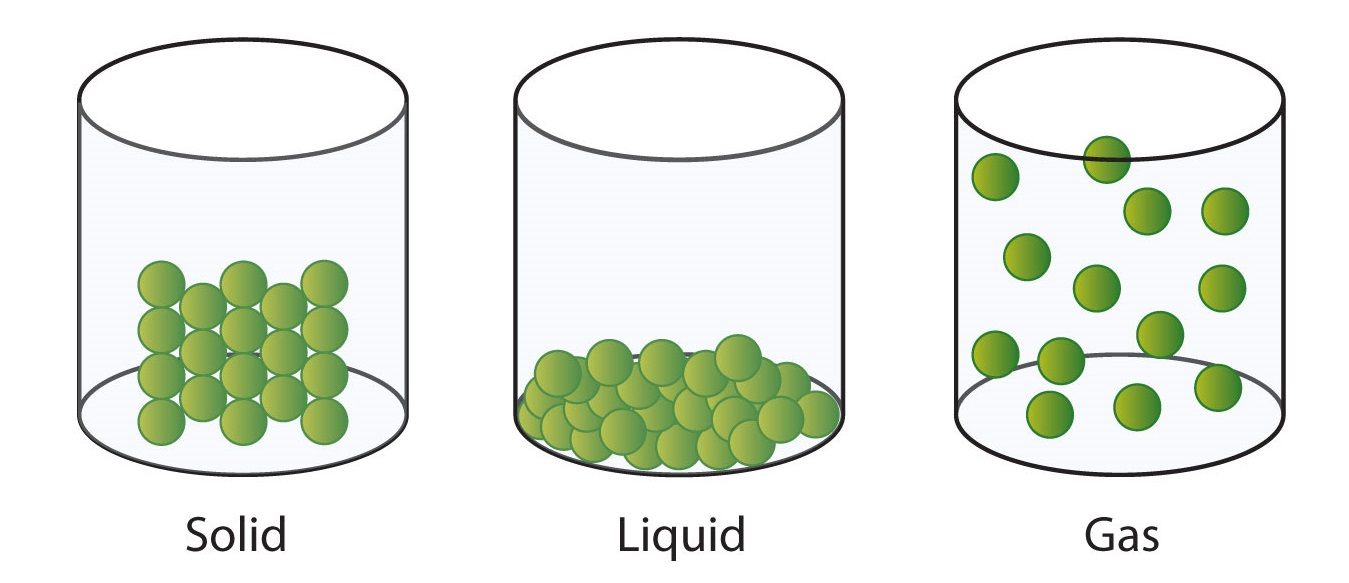
The alter from solid to liquid commonly does not significantly change the volume of a substance. However, the change from a liquid to a gas significantly increases the volume of a substance, by a factor of 1,000 or more than. Figure \(\PageIndex{three}\) shows the differences among solids, liquids, and gases at the molecular level, while Table \(\PageIndex{1}\) lists the dissimilar characteristics of these states.
| Characteristic | Solid | Liquid | Gas |
|---|---|---|---|
| shape | definite | indefinite | indefinite |
| book | definite | definite | indefinite |
| relative intermolecular interaction force | potent | moderate | weak |
| relative particle positions | in contact and fixed in place | in contact simply not fixed | not in contact, random positions |
What state or states of affair does each statement, describe?
- This state has a definite volume.
- This state has no definite shape.
- This state allows the individual particles to movement virtually while remaining in contact.
Solution
- This statement describes either the liquid state or the solid land.
- This argument describes either the liquid state or the gas state.
- This argument describes the liquid state.
What state or states of matter does each argument describe?
- This state has private particles in a fixed position with regard to each other.
- This land has individual particles far apart from each other in space.
- This country has a definite shape.
- Respond
-
a. solid
b. gas
c. solid
Looking Closer: Water, the Most Important Liquid
Earth is the only known body in our solar system that has liquid h2o existing freely on its surface. That is a good thing because life on World would not be possible without the presence of liquid h2o.
Water has several properties that make it a unique substance amidst substances. It is an splendid solvent; it dissolves many other substances and allows those substances to react when in solution. In fact, water is sometimes chosen the universal solvent considering of this ability. Water has unusually high melting and humid points (0°C and 100°C, respectively) for such a small molecule. The humid points for similar-sized molecules, such as marsh gas (BP = −162°C) and ammonia (BP = −33°C), are more than 100° lower. Though a liquid at normal temperatures, water molecules experience a relatively strong intermolecular interaction that allows them to maintain the liquid phase at college temperatures than expected.
Dissimilar most substances, the solid grade of water is less dense than its liquid form, which allows ice to float on water. The most energetically favorable configuration of HiiO molecules is one in which each molecule is hydrogen-bonded to four neighboring molecules. Owing to the thermal motions, this platonic is never achieved in the liquid, but when h2o freezes to ice, the molecules settle into exactly this kind of an arrangement in the ice crystal. This arrangement requires that the molecules be somewhat further autonomously than would otherwise exist the instance; as a consequence, ice, in which hydrogen bonding is at its maximum, has a more open structure, and thus a lower density than water.
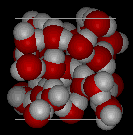
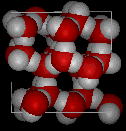
Here are three-dimensional views of a typical local structure of water (left) and ice (right.) Notice the greater openness of the ice construction which is necessary to ensure the strongest degree of hydrogen bonding in a compatible, extended crystal lattice. The structure of liquid h2o is very similar, but in the liquid, the hydrogen bonds are continually broken and formed because of rapid molecular motion. Because water ice is less dense than liquid water, rivers, lakes, and oceans freeze from the acme down. In fact, the water ice forms a protective surface layer that insulates the residuum of the water, allowing fish and other organisms to survive in the lower levels of a frozen lake or sea. If ice were denser than the liquid, the ice formed at the surface in cold weather would sink as fast every bit it formed. Bodies of water would freeze from the lesser up, which would be lethal for most aquatic creatures. The expansion of water when freezing as well explains why automobile or boat engines must be protected past "antifreeze" and why unprotected pipes in houses intermission if they are immune to freeze.
H2o besides requires an unusually large amount of energy to change temperature. While 100 J of energy volition alter the temperature of 1 yard of Fe by 230°C, this same corporeality of energy volition alter the temperature of 1 thou of H2O by only 100°C. Thus, water changes its temperature slowly as heat is added or removed. This has a major impact on weather, as storm systems like hurricanes can exist impacted by the amount of heat that ocean water can store.
Water's influence on the world around united states of america is affected by these backdrop. Isn't it fascinating that such a small molecule tin can have such a big bear upon?
Key Takeaway
- Solids and liquids are phases that have their own unique properties.
Definite Shape Of A Solid,
Source: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Basics_of_General_Organic_and_Biological_Chemistry_(Ball_et_al.)/08%3A_Solids_Liquids_and_Gases/8.02%3A_Solids_and_Liquids
Posted by: lightlebript1993.blogspot.com


0 Response to "Definite Shape Of A Solid"
Post a Comment